Doubt Cast on the Dutch Puberty Blocker Review
By Hermes Postma
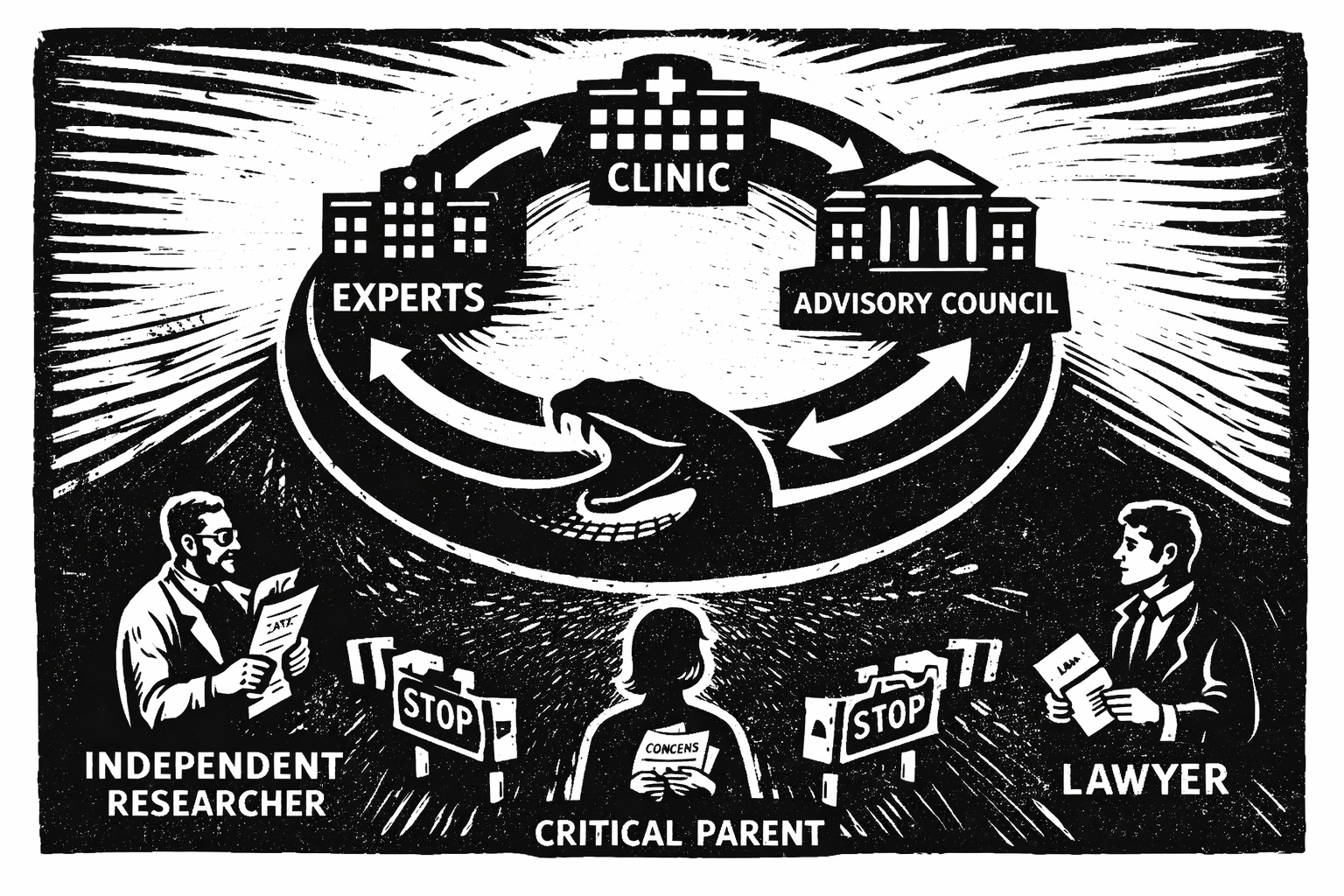
The Dutch are conducting their own review of puberty blockers, but will it be independent?
For more than two decades, the Netherlands has played a pivotal role in the global development of pediatric gender medicine. It was here that the Dutch Protocol was first formulated, providing the clinical rationale for prescribing puberty blockers to minors diagnosed with gender dysphoria. What began as a small, highly specialized practice in the Amsterdam clinic was subsequently promoted internationally as a cautious, evidence-based model and swiftly adopted across Europe and North America. Dutch institutions have not merely participated in the expansion of the use of puberty blockers; they have helped define their scientific and ethical foundations.
Today, however, the domestic climate in the Netherlands is less settled than this reputation suggests. While official guidelines and institutional statements continue to defend existing practice, there is growing unease among journalists, legal scholars, clinicians outside the specialist gender clinics, and an increasing number of parents. International developments, most notably the UK’s Cass Review (2024), the USA’s HHS Report (2025), and 16 other systematic reviews with identical conclusions, have intensified this reassessment, making it harder to dismiss concerns as marginal or ideological. Against this backdrop, Parliament’s request for an independent legal and medical evaluation of puberty blockers reflects a recognition that a practice so closely associated with national prestige now requires unusually robust scrutiny.
A Parliamentary Mandate Amid Growing Doubt
In December 2025, Dutch legal scholar Professor Lodewijk Smeehuijzen published a detailed critique of this process in the Netherlands Journal of Jurisprudence (Nederlands Juristenblad). His analysis focuses on the Dutch Health Council (Gezondheidsraad), which was tasked by Parliament with advising on the legal and medical legitimacy of prescribing puberty blockers to minors. Smeehuijzen’s concern is not only whether the Council’s conclusions will be sound, but whether the advisory process itself can credibly be regarded as independent.
The Health Council’s advice carries exceptional weight in the Dutch system and typically shapes national medical guidelines and health policy. In this case, it will strongly influence the ongoing revision of the Dutch Protocol, a framework that has guided youth gender medicine internationally for more than twenty years. The stakes are therefore high.
Parliament explicitly requested an independent assessment of whether the current practice complies with existing health law and accepted scientific standards for this very reason. According to Smeehuijzen, the Health Council’s advisory committee is poorly equipped to meet that mandate. Of its twelve members, six are directly or indirectly involved in prescribing puberty blockers or cross-sex hormones, and several are affiliated with the institutions that developed, implemented, and publicly defended the Dutch Protocol. Those same institutions would face significant reputational, professional, and potentially legal consequences if the practice were deemed unlawful or scientifically unsound.
Although some of these individuals are designated as “non-voting experts,” they nonetheless participate fully in deliberations and shape the framing of evidence. The Health Council’s own Code for the Prevention of Improper Influence due to Conflicts of Interest makes no distinction between voting and non-voting members. Smeehuijzen argues that this creates, at a minimum, the appearance of compromised independence. This appearance alone is sufficient to undermine public trust.
Regulatory Capture: Experts Assess Themselves
The article further stresses that this advisory request is not comparable to routine Health Council consultations. It combines a highly polarized international debate with an unusually small clinical field, meaning experts are effectively asked to assess their own practices. The reputational stakes are high for Dutch academic medical centers. The potential consequences include restructuring or suspending services. The review is retrospective and scrutinizes a practice that has been ongoing for more than thirty years. The explicit legal dimension of the inquiry also raises the prospect of future civil or disciplinary liability. According to Smeehuijzen, no recent Health Council process has as many compounding risk factors.
Despite Parliament’s request for legal evaluation, the committee includes only one legal scholar. That jurist has previously published on puberty blockers, off-label prescribing to minors, and the legal status of clinical protocols, and has co-authored work with a close colleague (also in the committee) who actively practices under the Dutch Protocol. Given the complexity of issues such as off-label drug use, informed consent by minors, children’s rights, and the normative force of clinical guidelines, Smeehuijzen concludes that the committee’s legal capacity is manifestly insufficient.
These concerns are underscored by a broader Dutch regulatory environment that, in Smeehuijzen’s view, is structurally ill-equipped for independent oversight. Access to underlying clinical data remains limited, off-label puberty suppression continues without a comprehensive research protocol, and care and research are routinely blurred by the same institutions providing treatment and producing the evidence base. External experts report professional and reputational risks associated with dissent, patient representation is dominated by trans advocacy organizations rather than minors, and policymakers rely heavily on information supplied by the institutions under review—a pattern Smeehuijzen characterizes as regulatory capture.
The implications extend beyond the Netherlands. The Dutch Protocol has long been presented internationally as a gold standard. Smeehuijzen’s analysis suggests that the safeguards meant to ensure independent evaluation of this model are failing precisely at a moment of heightened international scrutiny. His conclusion is clear: when medical interventions can irreversibly affect the physical, sexual, and reproductive development of minors, institutional distance between evaluators and practitioners is not optional but a requirement of the rule of law. Without visible independence, disciplinary breadth, and transparency, any reassessment of puberty blockers risks lacking legitimacy, regardless of its outcome.
Genspect publishes a variety of authors with different perspectives. Any opinions expressed in this article are the author’s and do not necessarily reflect Genspect’s official position. For more on Genspect, visit our FAQs.

