Dr Hilary Cass, WPATH and the Puberty Blocker Trial: Gender identity ideology undermining science.
By Peter Jenkins

NHS (UK) treads old ground in exploring ‘risks and benefits’ of puberty blockers for children.
This is a guest post from Peter Jenkins, a counsellor, supervisor, trainer and researcher in the UK and a member of Thoughtful Therapists. This article was previously published by Critical Therapy Antidote.
A major debate has broken out in the UK about the National Health Service (NHS) clinical trial into the use of puberty blocking medication for children. Following a judicial review into the Tavistock Gender Identity Development Service (GIDS) in 2020, the controversial unit was deemed ‘inadequate’ by a government health inspection agency, the Care Quality Commission, and subsequently closed down. Leading paediatrician Hilary Cass was appointed to lead a wide-ranging review of gender identity services for children and published a detailed report on the subject in 2024. Her review found the evidence for the use of puberty blockers, which are designed to prevent the development of secondary sexual characteristics in children experiencing gender distress, to be ‘remarkably weak’. In fact, rather than giving children under 16 ‘time to think’, the use of puberty blockers seemed to reinforce their wish to progress to cross-sex hormones and, later, surgical intervention. The use of puberty blockers in the UK was banned indefinitely in 2024. However, they are now set to be used again as part of a randomised controlled trial (RCT) involving 226 children as part of a complex research package. This move has reignited the fierce debate surrounding the use of puberty blockers. Critics are calling for the trial to end, deeming it unnecessary and unethical.
So, why this apparent turnaround in government health policy? In fact, Cass’ whole approach has always been to call for a robust, evidence-based approach to gender identity practice. This would be the case for any aspect of child healthcare. She called for the RCT as early as July 2022. However, a lot has happened in the meantime. The debate over so-called ‘gender identity affirming medicine’ has moved from apparently being just a niche, specialist medical topic to one of intense and growing wider public debate. Public awareness and scepticism about trans claims and entitlement seems to be growing, according to current polls (YouGov, 2025).
GIDS 1: Risks and Benefits of Puberty Blockers
To many, a major research programme investigating the risks and benefits of prescribing puberty blockers to children under 16 seems to be a significant step backwards. The reasons for setting up the trial partly lie in the poor track record of previous research into gender identity issues. Puberty blockers are extremely powerful drugs, technically defined as Gonadotropin-Releasing Hormone agonists (GnRHa). These have been used effectively in the past to treat precocious puberty and adult sex offending. A specialist clinic in the Netherlands pioneered their use with children seeking to undergo later medical transition. This became known as the Dutch Protocol (Delemarre-van de Waal, 2006). The Tavistock GIDS then adopted this model, originally as part of a limited research programme involving 44 participants. However, the original version of the research proposal (for what could be termed GIDS 1) was rejected by the first Research Ethics Committee (REC). This was on the grounds that the research design meant that there was no way to validate any of its findings without a control (i.e. placebo, or ‘no treatment’) group. Instead, the REC suggested “a trial which compared those who immediately started the intervention with those who had a delayed start” (Barnes, 2023: 55).
Undeterred, the GIDS simply took their proposal to another REC for approval. However, the results were ultimately inconclusive with regard to the effectiveness of puberty blockers in resolving gender dysphoria (Carmichael, 2021). A third of the small sample reported improvements in positive mood, a third reported no change, and a third reported negative changes in mood (Barnes, 2023: 371). This was hardly a stunning success by any measure.
However, the research programme had very quickly developed a dual purpose. The programme had formally started in 2011. Within a few years, however, “the referring of under-16s for puberty blockers was routine clinical practice at GIDS, as, remarkably, the service had not waited for the results of the trial before rolling the treatment out” (Barnes, 2023: 118). The research programme was thus arguably more effective in legitimising the wider use of puberty blockers, extending far beyond the small number of children formally enrolled in the original research protocol. This factor may also apply in the future to the randomised controlled trial. In fact, the applicants for the Pathway Trial have expressed an interest in increasing the size of their research sample beyond the current agreed limit of 226 patients (LCE REC, 2025: 2).
GIDS 2: Pathways Randomised Controlled Trial
The proposed new research programme is called ‘Pathways’. In many ways, it resembles the first puberty blocker trial. Indeed, it could perhaps more accurately be referred to as ‘GIDS 2’. One of the overarching aims of the research is “to determine the short/medium-term benefits and risks of GnRHa for puberty suppression” (KCL, 2025: 23). This phrasing is almost identical to that used in the original research submission made for GIDS 1 in 2011 (Barnes, 2023: 53). The research design differs in that GIDS 1 was an observational cohort study with no control group for comparison purposes. In fact, GIDS 2/Pathway has picked up and run with the original REC’s suggestion of a deferred treatment group for comparison purposes. This design is also closely related to a suggestion from the European Professional Association for Transgender Health (EPATH): “An alternative might be a waiting-list control group” (2025: 2). Thus, the GIDS 2/Pathways study is a randomised controlled trial that relies on a comparison between two groups receiving puberty blockers. The second group will start receiving puberty blockers one year after the first group. There is no placebo or ‘no treatment’ group for direct and sustained comparison. There is, therefore, no way to establish causality in a strict sense. The research will, no doubt, produce masses of data, including brain scans, but cannot, in its present format, produce definitive outcomes.
This raises major ethical problems, which critics have not been slow to point out. Considerable research already indicates that puberty blockers pose a significant risk of harm to patients. One systematic survey has reported that “Substantial evidence from peer-reviewed scientific studies, case studies, and clinical trials suggests that puberty-blocking drugs can negatively affect the skeleton, cardiovascular system, thyroid, brain, genitals, reproductive system, digestive system, urinary tract, muscles, eyes, and immune system” (Lesbians United, 2022: 16). Furthermore, “the best available evidence suggests that GnRH agonists have a number of irreversible or possibly irreversible effects on the human body” (Lesbians United, 2022: 14).
Risks of Harm from Puberty Blockers
In response, the research protocol for the GIDS 2/Pathways study acknowledges a host of possible risks to be covered in the informed consent process. These side effects are included in an eye-watering, extensive list of possible harms. These include depression and altered mood in children and anxiety, pain, sleep disorders and weight gain in adults (KCL, 2025: 75-6). However, the research protocol crucially fails to mention that many of these are potentially irreversible in nature, which is surely a significant consideration (Lesbians United, 2022: 14). There also seems to be a reluctance to acknowledge the full extent of the existing research on the proven harms of puberty blockers. This includes the irreversibility of certain types of harm, such as cognitive impairment (Biggs, 2022), and the known future risks, such as regret and detransition, as well as the increased risk of suicide in fully transitioned adults (Dhejne, 2011).
In narrow research terms, the case for clinical equipoise needed to justify the trial is therefore very weak. There is a clear admission by the research team regarding the risk of osteoporosis that ”studies show decreased age standardised bone mineral density with GnRHa” (KCL, 2025: 27 PJ: emphasis added). The research programme’s interest lies in “whether these changes are persistent after ceasing GnRHa and/or commencing on gender affirming hormones” (KCL, 2025: 27). This is hard to justify as a compelling argument for exposing participants to what is a known and acknowledged risk of further likely harm. Finding out whether this risk is short-term or long-term, and reversible or irreversible, is not at all an ethically sound reason for exposing vulnerable children to this grossly unacceptable degree of risk.
Influence of Unfalsifiable Gender Ideology
The research protocol appears to be constrained within a post-modernist, anti-scientific research paradigm. This is because it is centred on the concept of gender identity as an autonomous driver of social, medical and legal transition. Gender identity is par excellence an unfalsifiable belief, with no substantive agreed criteria or definition. Prescribing puberty blockers to children therefore amounts to providing medical treatment in order to comply with and endorse an unfalsifiable belief system held by the patient and/or parent. It is not at all equivalent to addressing a proven medical or psychiatric condition, despite the trial’s reliance on the ICD-11 diagnostic label of ‘gender incongruence’. Yet it is striking how spurious and misleading parallels are constantly drawn by its advocates between research into established medical conditions and research into the efficacy of puberty blockers. This is presumably intended to establish the legitimacy of the trial’s focus. Hence, we find comparisons of the trial to the “effort to develop and test new interventions for childhood cancers” (KCL, 2025: 26); or similarities to contrasting the treatment effects of “chemotherapy and radiotherapy” (Cohen, 2025: 2); or parallels with the need to establish ”the short-term benefits of stimulant medication in ADHD before examining longer-term effects” (KCL, 2025: 27).
The research protocol’s narrow focus on gender identity as the key driver of children’s gender distress and the potential means of resolving it bears a striking resemblance to the ideological language of the World Professional Association for Transgender Health (WPATH). WPATH is a hybrid trans activist body that has taken control of gender identity medicine in the US by popularising its Standards of Care, Version 8 (Jenkins, 2023a; Jenkins, 2023b). Its membership in the UK may be small, but its ideological influence over the terms of debate and discussion is remarkably resilient. Hence one of the FAQs for the research protocol refers to the participant goals of ‘body alignment with gender’ (KCL FAQs, 2025: 9). This phrase is curiously close to the stated aim of WPATH’s SOC 8 for its patients. This is “to better align their body with their gender identity” (Coleman et al, 2022: S31). There is also perhaps a further clue in the protocol’s frankly reductive and dismissive terminology used for psycho-social and psychological approaches i.e. as ‘non-endocrine intervention for CYP with gender incongruence’ (KCL, 2025: 22).
WPATH’s Politicised Approach to Research
In fact, WPATH’s strongly ideological approach to gender identity research suggests that it is a modern example of a ‘hyper-politicised approach to science’. In many ways this is akin to earlier approaches, i.e. “as arms of a particular political ideology” (Gordin, 2021: 30). Other examples would include the concept of ‘Aryan physics’ under the Nazi regime, Lysenkoism in genetic biology in Stalin’s USSR, and the eugenics movement during the twentieth century. Some of the major differences between a classic medico-scientific paradigm and a gender identity ideological paradigm (based on ideal-types) are briefly set out in the Table below.
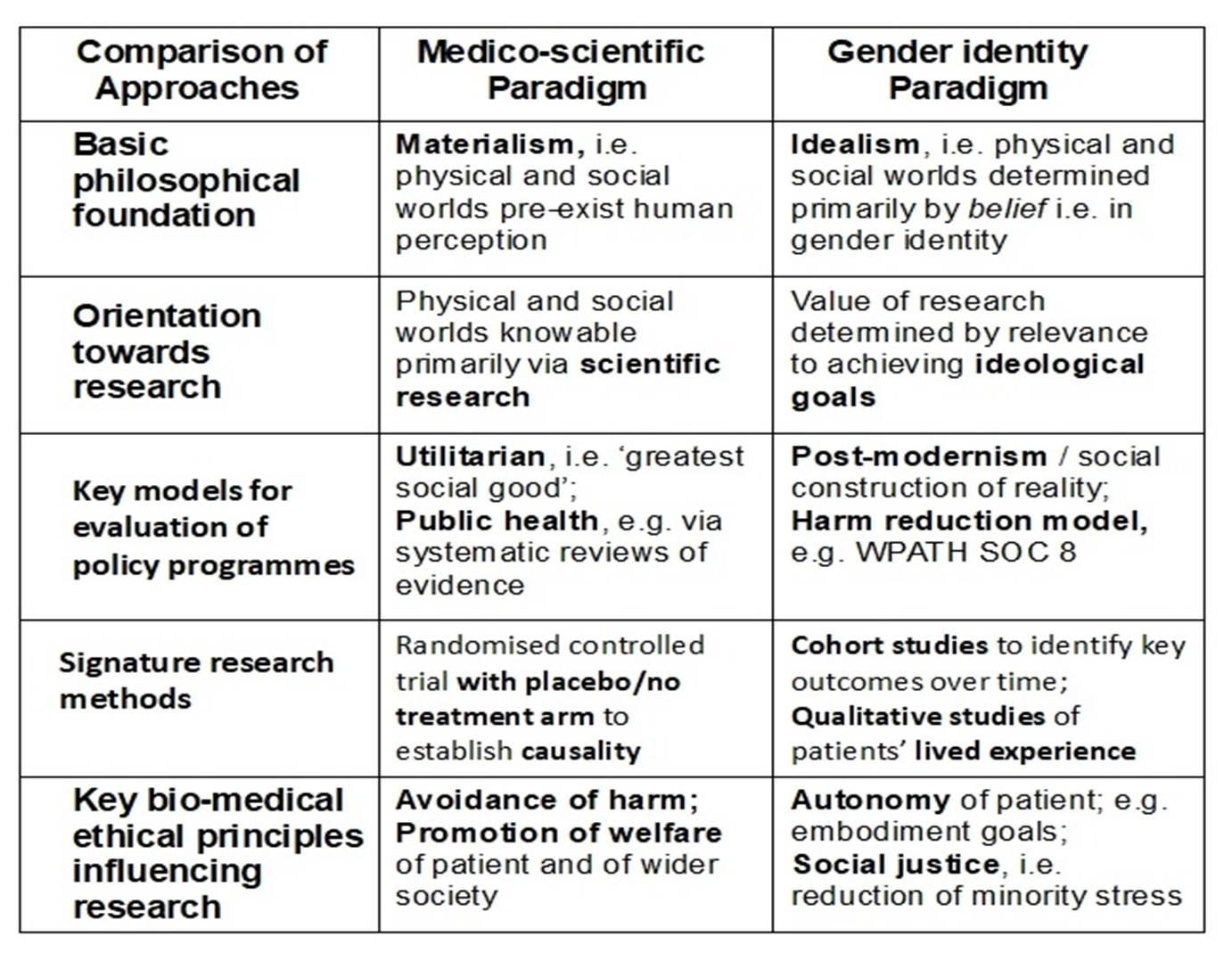
Table: Comparison of Medico-scientific and Gender identity Ideological paradigms
Probably the clearest expression of gender identity as a coherent ideology is to be found in WPATH’s Standards of Care Version 8 (Coleman et al, 2022). This version of the gender identity paradigm has clearly influenced the design and priorities of the puberty blocker trial. Both WPATH and the research protocol share a strong opposition to the use of a placebo or ‘no treatment’ group. For WPATH, this is argued on largely ethical grounds, as being both coercive and a denial of autonomous access to effective treatment for those allocated randomly to the placebo group (EPATH, 2024; McNamara, 2024). WPATH has apparently blocked the publication of its own commissioned systematic reviews on the grounds of their unfavourable findings. WPATH’s stance here would support the suggestion made in the Table above that the value of its own research is crucially determined by whether or not it contributes to the achievement of its wider ideological goals, namely the growth of gender identity ideology’s power, influence and the number of its adherents (DHHS, 2025: 166-170).
Trading Short Term Satisfaction for Longer-Term Harm?
The other parallel is perhaps less obvious. This is the central concept of ‘harm reduction’, which is at the heart of WPATH’s approach to gender identity affirming care: “SOC-8 supports…the value of harm reduction approaches” (Coleman, 2022: S6). Harm reduction is a reputable intervention where there is a convincing evidence base for its proper use. With WPATH, harm reduction becomes simply an overriding moral imperative. It variously justifies surgical intervention for eunuchs, or the use of breast binders for gender distressed girls. WPATH decries the use of delay, gatekeeping, prolonged assessment, or withholding of treatment as likely to cause harm to potential beneficiaries of medical care, in that it is likely to “to thwart an adolescent’s expression of gender diversity or assertion of a gender identity” (Coleman, 2022: S53; PJ: emphasis added). Harm reduction in this context too often seems to consist of a trade-off between offering vulnerable patients short-term improvements in their satisfaction levels, or in their immediate sense of well-being, as against the known risks of longer-term physiological or psychological damage.
It is more than concerning, therefore, to find the persuasive motif of harm reduction employed in the service of promoting the trial. Even Hilary Cass does not seem immune to this, raising the question in Parliament as to whether “it is better that they get their medication under careful clinical supervision rather than on the dark web?” (Hansard, 2025). The trial could therefore conceivably be justified, at least in part, on the grounds that it would provide safe and supervised access to puberty blockers. Notably, the same harm reduction argument was also advanced for the original GIDS 1 trial, when the equivalent concern voiced was that patients would go abroad unless puberty blockers were provided in some format by the GIDS (Barnes, 2023: 54).
Puberty Blockers RCT and the Hierarchy of Evidence
So where does the proposed trial sit within a wider hierarchy of research evidence on gender identity? Normally, randomised controlled trials sit close to the apex of such a hierarchy. However, Greenhalgh has suggested a necessary distinction needs to be made between RCTs with ‘definitive results’ and those with ‘non-definitive results’ (Greenhalgh, 1997). The proposed trial does involve random allocation to treatment groups, but without an independent placebo or ‘no treatment’ group. The trial cannot therefore establish causality on key issues, such as the reduction of bone density, or an increase in cognitive impairment, or the resolution of gender dysphoria. The following diagram suggests a possible hierarchy of research on gender identity, ranging from systematic reviews to self-report surveys.

References: 1 Cass, 2024; 2 NICE, 2020; 3 Cochrane, 2025; 4 Greenhalgh, 1997; 5 KCL, 2025; 6 de Vries, 2014; 7 Carmichael, 2020; 8 Hall, 2021; 9 Uyar, 2023; 10 Turban, 2020; 11 Cohen-Kettenis, 2011; 12 Ashley, 2019; 13, GEO, 2018; 14 James, 2016; 15 OpenMD, 2025.
According to this schematic representation, much current research into gender identity tends to cluster around the method of cohort studies, with a marked absence of classic ‘gold standard’ randomised controlled trials, capable of producing definitive results. Clearly, some important medico-scientific research can be done on the topic of gender identity. It is, after all, possible to carry out robust research into belief systems, such as politics, religion, or gender identity, without necessarily subscribing to the validity of the underpinning belief itself. While the GIDS 2/Pathways trial will inevitably produce reams of data, it is not capable of establishing causality because of the way it has been set up, i.e. without a placebo or ‘no-treatment’ group. So when the press reports that “A Department of Health and Social Care spokesman said ‘This trial will help provide the evidence that is lacking’”, this seems extremely unlikely to be the case (Simpson, 2026).
Judicial Review to Test Questionable Ethics
The attempt to stop the UK National Health Service trial of puberty blockers has now moved decisively into the legal arena. James Esses set up an online petition against the proposed trial of puberty blockers, which were largely banned in the UK in 2025. The petition rapidly reached 140 thousand signatures, meaning it had passed the threshold required for debate in Parliament. This debate is planned for March 8, 2026. However, the Department of Health and Social Care (DHSC) has rejected the petition’s arguments, using a mix of administrative, procedural and operational reasons to justify the trial.
Ultimately, the DHSC is sheltering behind selective reference to the Cass Review. It seems to want to avoid opening up yet again the risky debate about the highly questionable ethics of subjecting children to known and likely harms of blocking the normal puberty of vulnerable child patients. So, according to the DHSC, “We are following the expert advice of the Cass Review to establish a clinical trial to determine the relative benefits and harms of puberty suppression in young people with gender incongruence.”
In response, James Esses then launched a judicial review on February 6, 2026 against the Department of Health and Social Care over its decision to run the controversial randomised controlled trial of puberty blockers for gender questioning children. According to James, “We gave the government every opportunity to halt the monstrous puberty blockers trial.”
Judicial review is a legal process to test whether government policy is both reasonable and following due process. This legal action is similar to the successful judicial review brought by detransitioner, Keira Bell, and others, against the Tavistock Gender Identity Development Service (GIDS) back in 2020. This was to review the GIDS’ policy of obtaining consent from children as young as 10 years of age to puberty blockers.
James, a trainee therapist and barrister, has previously brought successful legal challenges against both Metanoia, a psychotherapy training body, and its parent body, the United Kingdom Council for Psychotherapy (UKCP), for dismissing him from his therapy course after raising safeguarding issues. He reached an apology and out of court settlement with Metanoia and the UKCP in 2024. This track record of successful legal action so far might just be making the DHSC a tad nervous, as it dusts off its legal arguments for the forthcoming court case over the puberty blocker research trial.”
Summary
In many ways, GIDs 2/Pathways is following closely in the footsteps of its predecessor, i.e. GIDS 1. Both have diffuse and unrealistic overarching goals of exploring the nature and extent of the risks and benefits of puberty blockers for children experiencing gender distress. While adverse side effects are identified for the purpose of obtaining informed consent, the serious risks of major longer-term harms (regret/detransition, irreversibility, enhanced adult suicide risk) appear not to be clearly underlined. The reliance on parental consent may meet minimum legal standards for medical research trials, but is fraught with difficulty in practice (this key issue is discussed in more detail in Jenkins, 2026). The research protocol shares with WPATH a core resistance to employing a placebo or ‘no treatment’ group. This design feature is essential in order to establish causality, rather than simply produce yet more inconclusive findings. Hilary Cass, a strong advocate of the GIDS 2/Pathways trial, also shows similarity to WPATH’s use of a harm reduction model as a persuasive rationale for the trial, but without recourse to detailed and convincing evidence.
Puberty blockers, a dangerous and off-label drug, were originally brought in to the NHS by the back door via GIDS 1, based on the deeply flawed Dutch Protocol. There is now a bold attempt to bring puberty blockers back into wider use, but this time via the front door of the GIDS 2/Pathway trial. The latter’s RCT trial design clearly owes more to gender identity ideology than to straightforward medico-scientific research.
In political terms, the puberty blocker trial is an obvious sop to an aggrieved and vocal trans lobby. In research terms, this is an unnecessary exploration of the wider extent of harms which are already known and well-documented elsewhere, and which is set firmly within an unquestioned paradigm about gender identity and the case for medical transition. In medical terms, it is a fatal concession to the power of unbridled consumer demand for an off-label and dangerous medicine for vulnerable children, in order to buttress a belief system lacking any foundation in biological reality.
Non-electronic references:
Barnes, H. (2023) Time to think: The inside story of the collapse of the Tavistock’s Gender Service for Children. Swift: London.
Gordin, M. (2021) On the fringe: Where science meets pseudoscience. Oxford University Press: Oxford.
Acknowledgement:
Published with the permission of Critical Therapy Antidote; also reposted in Multi-Level Mailer.

Peter Jenkins is a counsellor, supervisor, trainer and researcher in the UK. He has been a member of both the BACP Professional Conduct Committee and the UKCP Ethics Committee. He has published a number of books on legal aspects of therapy, including Professional Practice in Counselling and Psychotherapy: Ethics and the Law (Sage, 2017).
Genspect publishes a variety of authors with different perspectives. Any opinions expressed in this article are the author’s and do not necessarily reflect Genspect’s official position. For more on Genspect, visit our FAQs.

